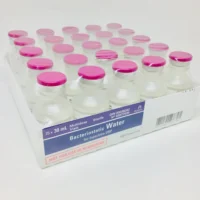
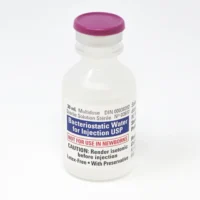
Hospira Bacteriostatic Water 30mL | Pfizer Canada Supply
£24.99
59,467 GBP/BTC
CoinGecko 14:45:27
330.11 GBP/XMR
CoinGecko 14:45:27
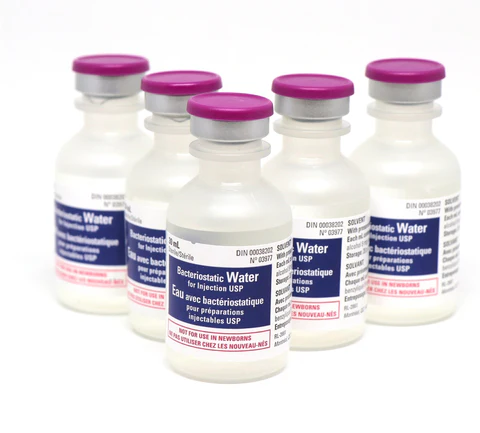
Hospira Bacteriostatic Water 30mL. Sterile, multi-dose Pfizer vial with 0.9% benzyl alcohol. Order online with fast UK delivery.
Back-order pricing live.
Available on back-order
Description
Hospira Bacteriostatic Water 30mL | For Research Use Only
Hospira Bacteriostatic Water is a pharmaceutical-grade sterile solution manufactured by Pfizer (Hospira), supplied strictly for research and laboratory purposes only. This product is not intended for use in humans or animals.
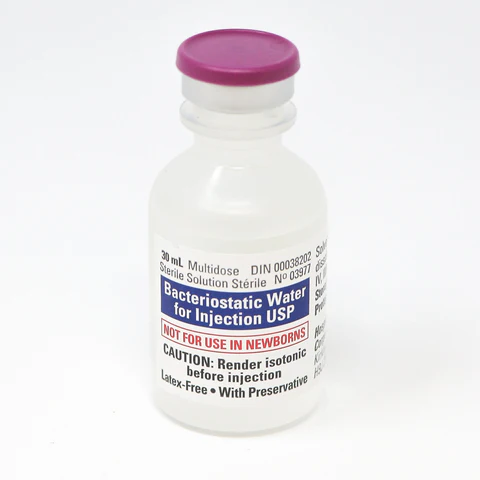
Each 30mL multi-dose vial contains sterile water with 0.9% benzyl alcohol as a bacteriostatic preservative, enabling multiple withdrawals from a single vial while maintaining sterility between uses making it a reliable choice for research environments requiring a contamination-resistant aqueous solution.
Product Specifications:
Manufacturer: Pfizer Canada (Hospira)
Volume: 30mL multi-dose plastic vial
Preservative: 0.9% benzyl alcohol
Sterile and non-pyrogenic – pH 5.7 (range 4.5–7.0)
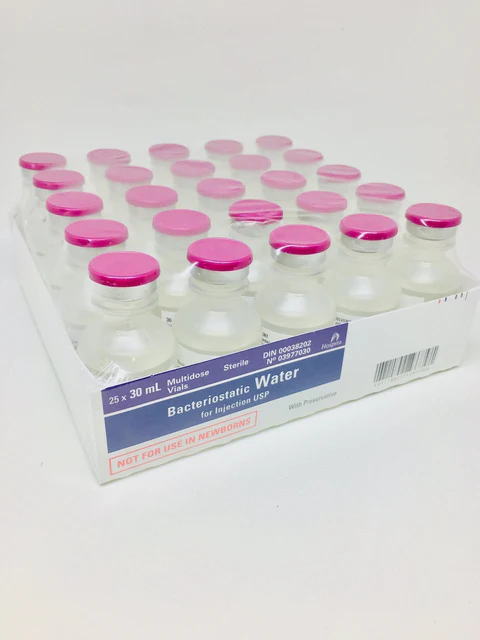
Available individually, in a 5-pack, or case of 25
Unsure what you need, check out our buyer’s guide.
Bacteriostatic Water and Bacteriostatic Saline are commonly used research diluents, often searched for under alternative terms such as bac water, BAC, and mixing solution. Below are answers to common questions about Bacteriostatic Water, Bacteriostatic Saline, Sterile Water, storage, stability, and common naming variations.
Bacteriostatic Water & Bacteriostatic Sodium Chloride FAQ
What is Bacteriostatic Water?
Bacteriostatic Water is a sterile water solution containing 0.9% benzyl alcohol as a preservative to help inhibit bacterial growth. It is commonly used for dilution and reconstitution in research settings where appropriate instructions are provided. On product pages, the correct term is Bacteriostatic Water, although some customers also search for it using other names.
What are other terms for Bacteriostatic Water?
Bacteriostatic Water is often searched for using terms such as bac water, BAC water, BAC, bacteriostatic mixing water, and mixing solution. These are common alternative search terms, but Bacteriostatic Water remains the most accurate and professional product name.
Is Bacteriostatic Water the same as bac water or mixing solution?
Yes, many people use terms like bac water, BAC water, or Bacteriostatic Water for Injection when referring to Bacteriostatic Water. These are common alternative search terms, while Bacteriostatic Water is the clearest product term for product pages and technical descriptions. Some researchers may also recognise major branded references such as Pfizer or Hospira labelling, but Bacteriostatic Water should remain the primary term.
What is the difference between Bacteriostatic Water and Sterile Water?
The main difference is that Bacteriostatic Water contains a preservative, while Sterile Water does not. The preservative helps inhibit bacterial growth after opening, which can make Bacteriostatic Water suitable for certain multi-use research applications where appropriate. Sterile Water is generally preservative-free and is more commonly associated with single-use situations.
How long does Bacteriostatic Water last after opening?
In our research environment, Bacteriostatic Water has been observed to remain stable for 4 to 8 weeks after opening. Actual stability depends on storage conditions, handling, and the researcher’s own risk profile. For research use, we recommend using Bacteriostatic Water within 6 weeks of opening.
How should Bacteriostatic Water be stored after opening?
Bacteriostatic Water should be stored according to the product label and kept in a clean, dry environment. Always inspect the vial before use and do not use it if the solution appears cloudy, discoloured, damaged, or otherwise compromised. Good storage and handling practices are important in supporting product quality during research use.
Important Notice:
This product is supplied for research purposes only. It is not approved, intended, or labelled for use in humans or animals. Researchers are responsible for ensuring compliance with all applicable regulations in their jurisdiction.
Additional information
| Weight | 115 g |
|---|---|
| Dimensions | 34 × 34 × 72 mm |
| Brand | Pfizer, Hospira |
| Volume | 30mL |
| Vial Type | Multi-dose |
| Preservative | Benzyl Alcohol, BA |
| Concentration | 0.9% |
| pH Range | 4.5 – 7.0 |
| Sterility | Sterile, Non-pyrogenic |
| Intended Use | Research & Laboratory Only |
| Pack Size | Individual, 5-Pack, Case of 25 |
| Country of Origin | Canada, CA |
| Manufacturer | Pfizer Canada ULC |
| Storage Conditions | Store at controlled room temperature out of light. |
| Colour | Clear |
| Container Material | Plastic, Polyolefin, Ethylene, Propylene |
| Estimated Back Order Lead Time | 6-12 Weeks or >12 Weeks |
Quick Comparison
| Hospira Bacteriostatic Water 30mL | Pfizer Canada Supply remove | Hospira Bacteriostatic Sodium Chloride 20mL | Pfizer Canada Supply remove | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | Hospira Bacteriostatic Water 30mL | Pfizer Canada Supply remove | Hospira Bacteriostatic Sodium Chloride 20mL | Pfizer Canada Supply remove | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Image | 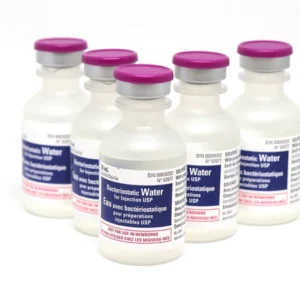 | 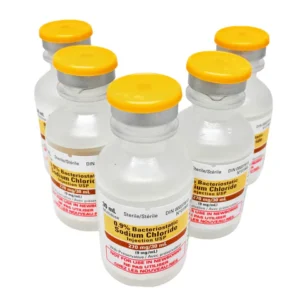 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Price | £24.99 | Original price was: £32.99.£27.95Current price is: £27.95. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additional information |
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Add to cart |